EARLY WARNING DETECTION
Tutivia is a precise multi-gene RNA signature test, developed through AI techniques that provides an early warning blood-based assay that can detect all types of acute rejection, preceding traditional markers.
Clinicians can use this test for both high and low-risk individuals, from the first week post-transplant, through subsequent follow-ups.
Providing proactive insights.
Catching a patient’s risk of acute rejection early may enable a patient’s clinical care team to proactively move to help protect the transplanted kidney.
Giving new meaning to peace of mind.
With a high odds ratio of 5.74, patients with an elevated Tutivia risk score are nearly 6x more likely to experience acute rejection events compared to a low risk Tutivia score.
- Acute antibody mediated rejection
-
T cell mediated rejection.
- Mixed rejection
- Borderline
- Subclinical

Tutivia Benefits:
Tutivia can be used as early as the first week after transplant and thereafter throughout its life, to detect the risk of rejection.
- With 85% specificity, Tutivia ensures highly accurate results by minimizing false positives, giving you confidence in accurate results and reducing unnecessary biopsies.
- With 83% sensitivity, Tutivia identifies Indviduals with a high risk of rejection within the first 60 days post-transplant.
Frequently Asked Questions
Verici Dx welcomes all insurance plans. The test is covered for use in Medicare patients once criteria are met.
Verici Dx is committed to the goal of helping all patients have successful outcomes after a kidney transplant.
To mitigate financial burdens, Verici Dx has developed a cost-sharing protocol. Call +1 (833) 243-4552 for more information.
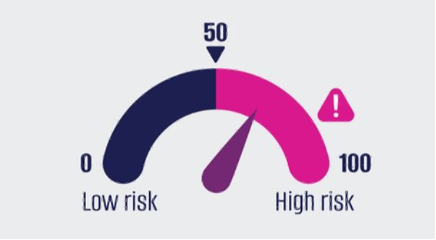
The Tutivia risk score has been designed to support actionable clinical decision-making through a clear, easy-to-interpret scoring scale.

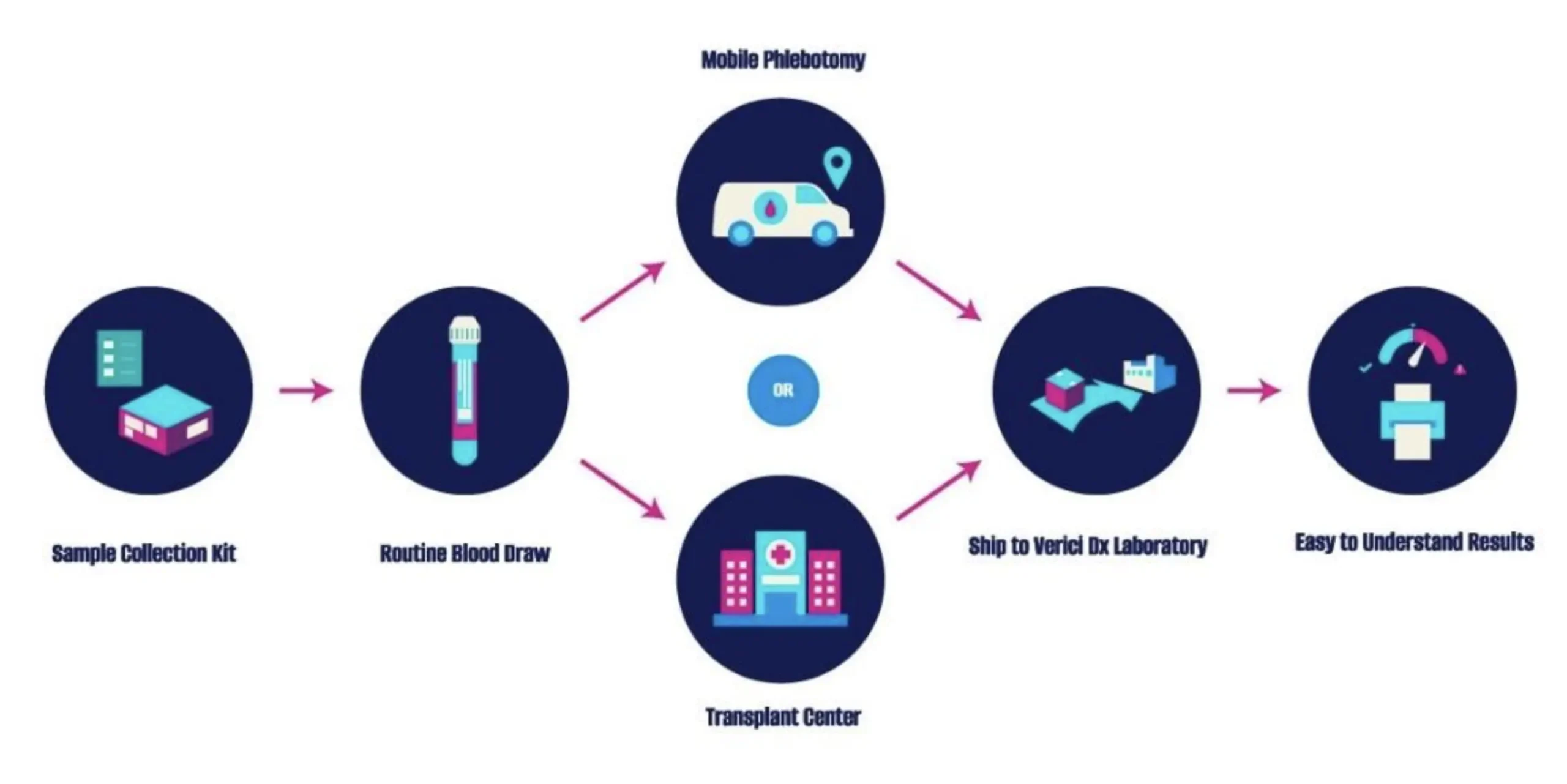
The transplant team determines the right time to perform the test. The test will likely be done in clinic or can be drawn through our mobile phlebotomy service. It is then sent to the Verici Dx lab for blood sample analysis. The result from the sample will then be sent back to the transplant team.
Tutivia can be used as early as the first week post-transplant. In the validation study, Tutivia was used both for-cause at any time and for routine surveillance at months 1, 3, 6, 12, and 24.
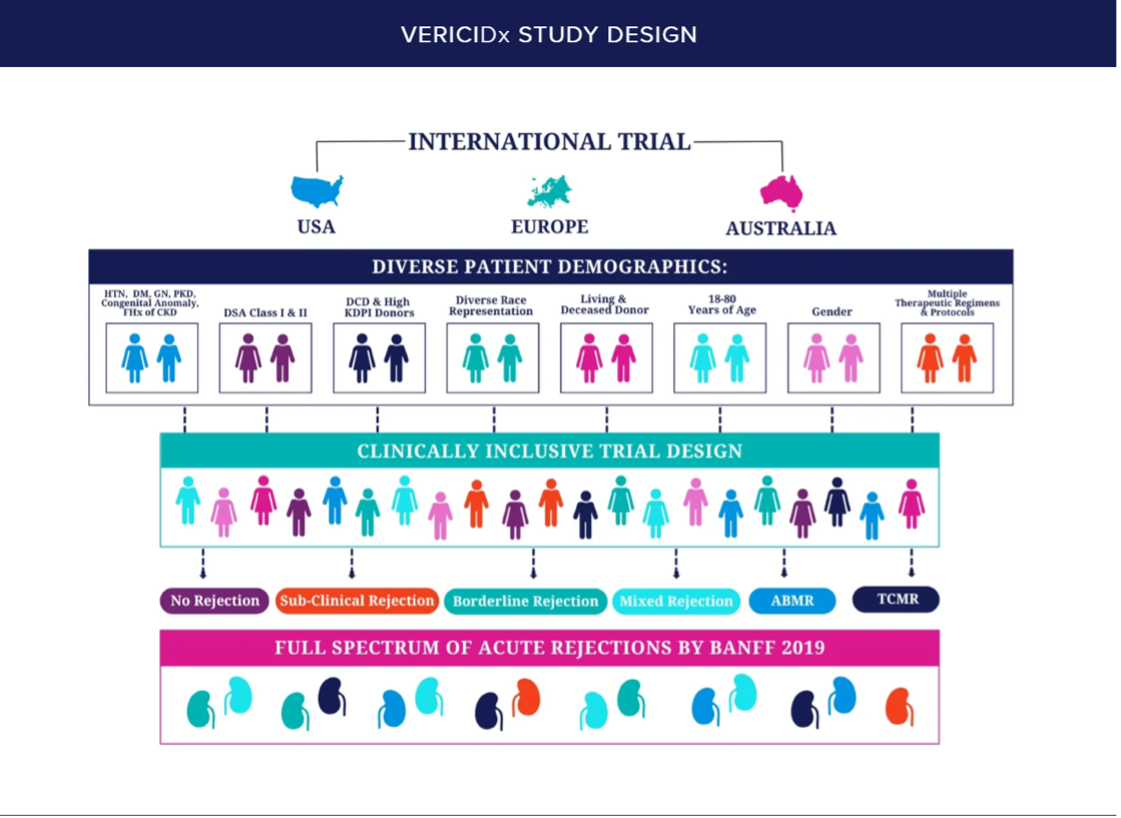
The Tutivia study included a very inclusive and diverse study group, designed to mirror the patient population most transplant centers serve. Contact us at +1 (833) 243-4552 to discuss with a dedicated specialist whether your patient is an appropriate candidate for Tutivia
The test does not measure anything that would be affected by food or medications. Currently there are no restrictions prior to having blood drawn to perform the test.